Treating Lung Metastases with Image-Guided Ablation
In This Article
- Thermal ablation offers a minimally invasive treatment option for lung metastases, particularly in patients who cannot tolerate surgical resection or radiation
- Thermal ablation techniques eradicate tumors with either extreme heat or extreme cold delivered through percutaneous applicators
- Image guidance ensures minimal damage to tissue surrounding the tumors, and pulmonary function is not affected
Lung metastases are often treated with surgical resection or radiation. However, for some patients, surgery and radiation are not options due to the extent of metastatic disease or comorbidities including limited respiratory function, advanced age or prior radiation therapy. For these patients, image-guided ablation represents an alternative approach.
Subscribe to the latest updates from Radiology Advances in Motion
Image-guided ablation treats metastases by applying thermal energy to tumors through percutaneously inserted probes. Skin incisions are only 3 mm wide, similar to a percutaneous needle biopsy. Positioning of the probes is typically guided by computed tomography. Once probes are in place, energy is released, which results in temperature changes that induce cell death in and around a tumor. Ablation techniques can use either extreme heat (radiofrequency and microwave ablation) or extreme cold (cryoablation).
A number of studies have established the safety and efficacy of image-guided ablation for lung metastases measuring less than 2 cm and have highlighted advantages compared with surgical resection and radiation therapy. In a review article in Seminars in Thoracic and Cardiovascular Surgery, the researchers explain why patients can undergo lung tumor ablation multiple times without impacting pulmonary function tests. The reason is the minimal damage to healthy lung tissue surrounding the tumor or parenchymal sparing. There is no set maximum number of lesions that can be ablated if a probe can be safely placed within each lesion using the percutaneous approach. Lesions can be widely distributed through one or both lungs. Ideal candidates have five or fewer lesions measuring less than 2 cm.
Another advantage of ablation is the short and minimally invasive nature of this treatment: Only one session is required per lesion, followed by an overnight hospital stay for observation. In addition, image-guided ablation does not preclude surgical resection or radiation therapy at a later stage. In other words, no treatment options are taken off the table.
Radiofrequency and Microwave Ablation of Lung Metastases
The best-known ablation modality is radiofrequency ablation (RFA). RFA destroys tumors using electromagnetic waves of 400 to 500 kHz. The electromagnetic waves produce frictional motion of electrons near the tip of the electrode, which in turn generates heat. Temperatures higher than 60 degrees Celsius—which can be achieved with the technique—kill cells instantly. Radiofrequency ablation is considered low risk and is well tolerated by patients.
In an Annals of Oncology article, researchers reported the efficacy of RFA for the treatment of lung metastases. They assessed overall survival and disease-control rates in 566 patients with 1,037 lung metastases. The researchers reported a five-year overall survival rate of 51%—only 2.5% lower than the best results obtained with surgery—and three- and four-year disease-control rates of 53.7% and 44.1%, respectively. They attributed the latter finding in part to the possibility of retreatment with the minimally invasive procedure.
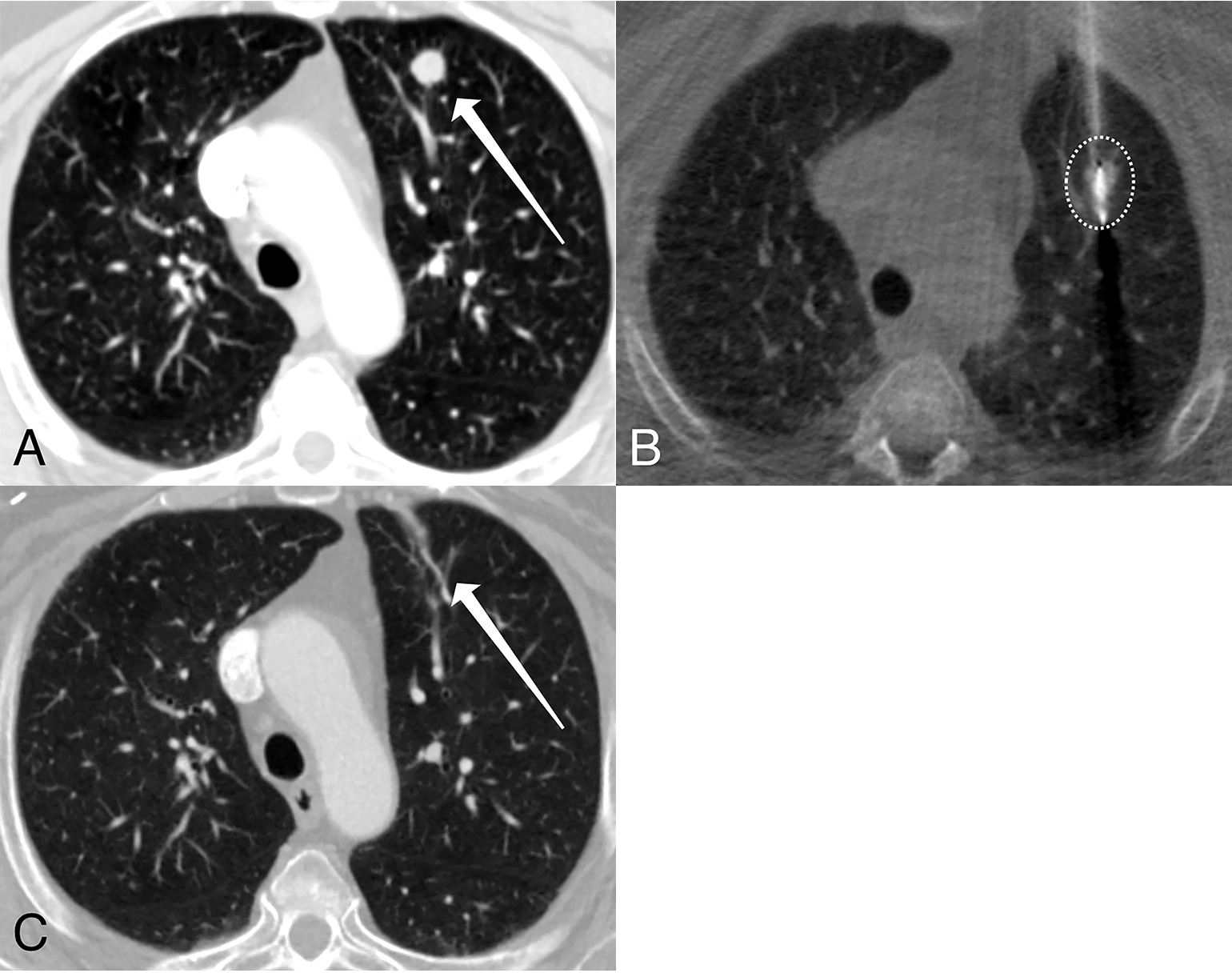
Figure 1
Microwave ablation of a lung metastasis. Serial computed tomography images of a 74-year-old female with metastatic endometrial cancer demonstrate an 11-mm left upper lobe solid lung nodule (panel A, arrow) prior to treatment. An image acquired during treatment (panel B) demonstrates a single ablation probe positioned within the lung metastasis; the white circle indicates the treatment zone. Six months following the ablation (panel C), only a small scar remains (arrow). Images courtesy of Florian J. Fintelmann, MD.
However, microwave ablation continues to replace RFA. Microwave ablation is also heat-based but uses higher frequency electromagnetic waves (900 to 2,450 MHz), which results in greater energy deposition compared with RFA. As a result, the temperature rises more quickly, leading to shorter procedure times. In addition, microwave ablation can achieve larger treatment zones than RFA. This larger heating zone of microwave ablation allows for more-uniform tumor destruction and targets measuring up to 3 cm.
Cryoablation of Lung Metastases
In contrast to RFA and microwave ablation, cryoablation induces cell death with extreme cold temperatures of below minus 50 degrees Celsius. The release of pressurized argon gas within the applicator and subsequent thawing results in intracellular and extracellular ice, which disrupt cell membranes and ultimately kill the cells. Because the ice can be visualized in real time, radiologists can monitor the size of the treatment zone during the procedure.
In 2015, the results of a prospective, multicenter study of cryoablation for lung metastases measuring up to 3.5 cm were reported in the Journal of Thoracic Oncology. Forty patients with 60 lung metastases were treated during 48 cryoablation sessions. The researchers found a striking 94.6% control rate at 12 months and noted that the treatment was generally well tolerated. They also noted that cryoablation offers advantages compared with RFA and microwave ablation. For example, cold creates an instant analgesic effect, which minimizes procedural pain, especially during the treatment of pleural and chest wall lesions, as described here.
Patient Evaluation Prior to Ablation of Lung Metastases
Multidisciplinary evaluation, including oncology, radiation oncology and thoracic surgery, is key to optimize care for an individual patient. Following a referral, patients at Mass General are seen by a subspecialized interventional radiologist skilled in lung ablation. Alternatively, the radiologist joins a clinic visit with the referring oncologist or thoracic surgeon. Needle biopsy is recommended prior to ablation to establish a tissue diagnosis, unless metastatic disease to the thorax has already been established with prior resection.
Scheduling
Lung ablations are performed by the Division of Thoracic Imaging and Intervention in the Department of Radiology at Massachusetts General Hospital in Boston. Orders can be placed in Epic (inside the Partners network) or Physician Gateway (outside the Partners network) or by calling 617-724-4254.
References
- Venuta F, Rolle A, Anile M, et al. (2010). Techniques used in lung metastasectomy. J Thorac Oncol 5(6 Suppl 2): S145-50.
Visit the Division of Thoracic Imaging and Intervention
Explore research in the Division of Thoracic Surgery