Local NAMPT Inhibition Enhances Checkpoint Immunotherapy for Glioblastoma
Key findings
- The characteristic immunosuppressive microenvironment of glioblastoma has made immunotherapy challenging
- In this study, intratumoral delivery of a nicotinamide phosphoribosyltransferase (NAMPT) inhibitor via polymer microparticles resulted in upregulation of immune checkpoint PD-L1, recruitment of T cells and M2-to-M1 macrophge polarization
- Local NAMPT inhibition followed by PD-1 inhibition prolonged survival in mice with glioblastoma whereas both monotherapies had limited activity
Subscribe to the latest updates from Oncology Advances in Motion
Cancer cells depend on nicotinamide adenine dinucleotide (NAD+), a co-enzyme that enhances glycolysis, to fuel their proliferation. Pharmacological inhibition of nicotinamide phosphoribosyltransferase (NAMPT) depletes NAD+ levels, suppressing cancer cell proliferation.
Recent evidence suggests NAMPT inhibitors also modulate normal immune cells within the tumor microenvironment. Those findings are relevant to glioblastoma because although immune cells represent up to 50% of glioblastoma volume, they typically suppress anti-tumor immunity.
However, systemic NAMPT inhibitors have significant adverse effects. In previous work, Massachusetts General Hospital researchers loaded a NAMPT inhibitor into polymer microparticles and applied them directly to glioblastoma, allowing sustained local release.
Extending that research, Daniel P. Cahill, MD, PhD, neurosurgeon at Mass General and neurosurgical oncologist at the Mass General Cancer Center, Hiroaki Wakimoto, MD, PhD, and Ming Li, MD, of the Mass General Cancer Center, and colleagues demonstrated that a microparticle-delivered NAMPT inhibitor reversed the immunosuppressive state of the glioblastoma microenvironment in mice. In Cancer Research, they explain how they exploited this finding to create a new combination immunotherapy for glioblastoma.
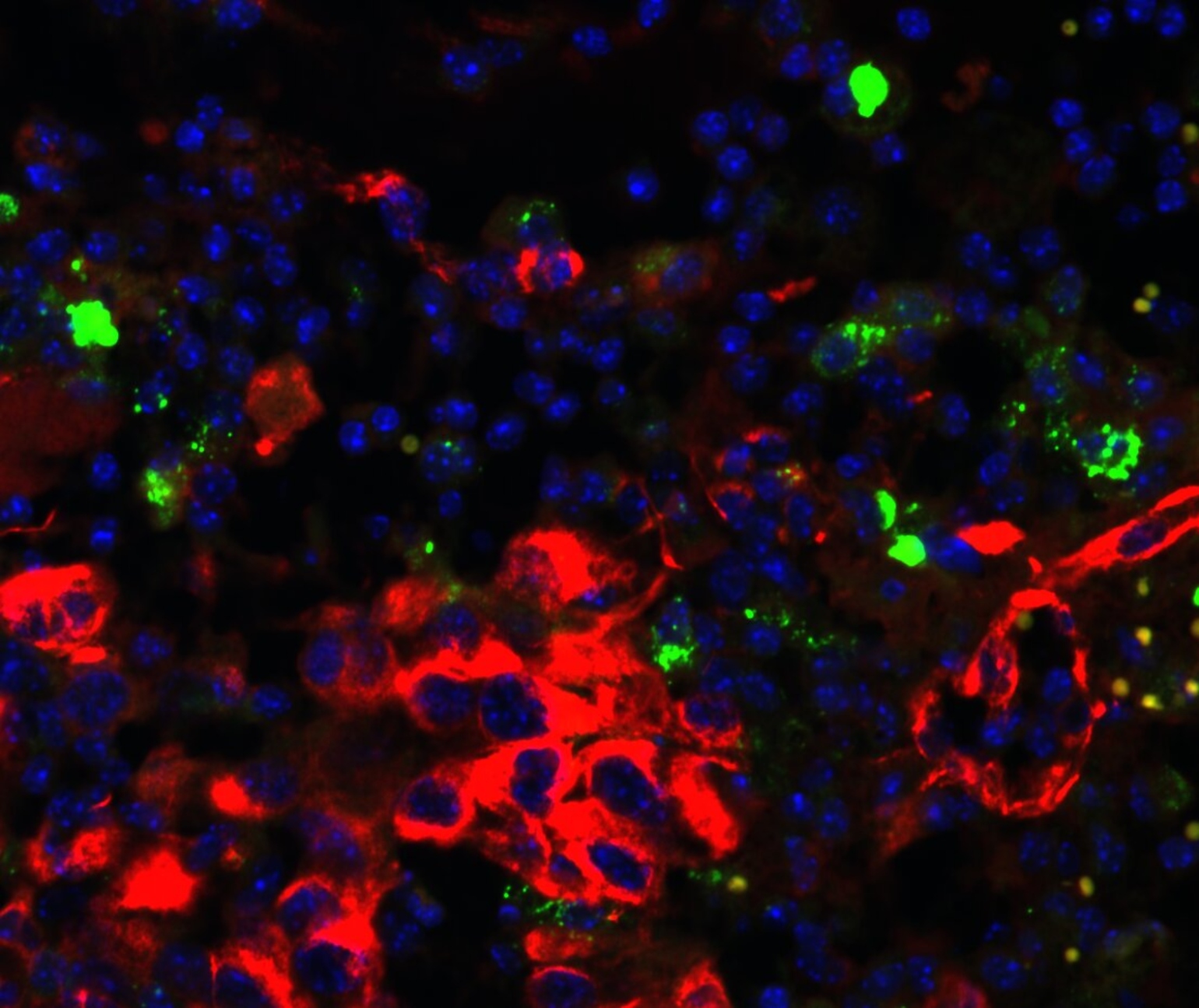
Figure 1
Immunofluorescence analysis showing the upregulation of PD-L1 (green) in murine glioblastoma (red, tumor cells) after microparticle-mediated local delivery of NAMPT inhibitor.
Immunologic Changes
The researchers injected microparticles containing GMX1778, an NAMPT inhibitor, into brain glioblastomas in mice. They observed dramatic changes in the immune cells infiltrating the tumors:
- Immune checkpoint PD-L1 was upregulated
- CD3+, CD4+ and CD8+ T cells were recruited
- Macrophages with an M1 (inflammatory and anti-tumoral) phenotype were increased and those with an M2 (anti-inflammatory and pro-tumoral) phenotype were significantly decreased
Combination Therapy
The team then injected GMX1778 microparticles into mice, followed by systemic PD-L1/PD-1 blockade. Upregulation of PD-L1 was no longer observed, and the combination significantly extended survival compared with GMX1778 or PD-L1/PD-1 inhibition alone. Intriguingly, one animal that received the combination became tumor-free. Treatments were well tolerated (no significant loss of body weight).
The known toxicities of systemic NAMPT and anti-PD-1 inhibitors in humans could additively increase the risk of adverse events after this combination therapy. Microparticle delivery may circumvent those problems.
view original journal article Subscription may be required
Learn more about research in the Mass General Cancer Center
Refer a patient to the Mass General Cancer Center