Activating CD4+ T Cells at Benign Nevi Might Prevent Melanoma
Key findings
- In this study, Massachusetts General Hospital researchers observed that patient-derived benign nevi (common moles) implanted into immunodeficient mice were spontaneously rejected
- CD4+ T helper 1 cells resident in the nevi were responsible for the rejection; ordinarily those cells are held in check by regulatory T cells (Tregs)
- As confirmation, nevus rejection was prevented when Treg activity was enhanced by administering low-dose human interleukin-2 or CD4+ T helper 1 cell function was blocked by a human leukocyte antigen II–blocking antibody in the xenografts
- When human-derived melanoma cells were introduced to a rejected nevus site in the xenografts, melanoma growth and metastasis were significantly blunted, illustrating the potential of anti-nevus T-cell immunity to protect against melanoma
Approximately 33% of melanomas begin in benign nevi. Shadmehr (Shawn) Demehri, MD, PhD, director of the High Risk Skin Cancer Clinic of the Center for Cancer Research at Massachusetts General Hospital, PhD candidate Erik B. Schiferle, and colleagues have determined it may be possible to harness the immune response to these nevi to prevent and treat melanoma. Their report appears in Science Advances.
Subscribe to the latest updates from Oncology Advances in Motion
Cytotoxic Immunity and Nevus Rejection
The researchers transplanted common melanocytic nevi from 33 human volunteers onto severely immunodeficient mice. Unexpectedly, 82% of the transplanted nevi were spontaneously rejected. Additional experiments demonstrated that activation of CD4+ T helper 1 cells in the nevi was responsible for the rejection.
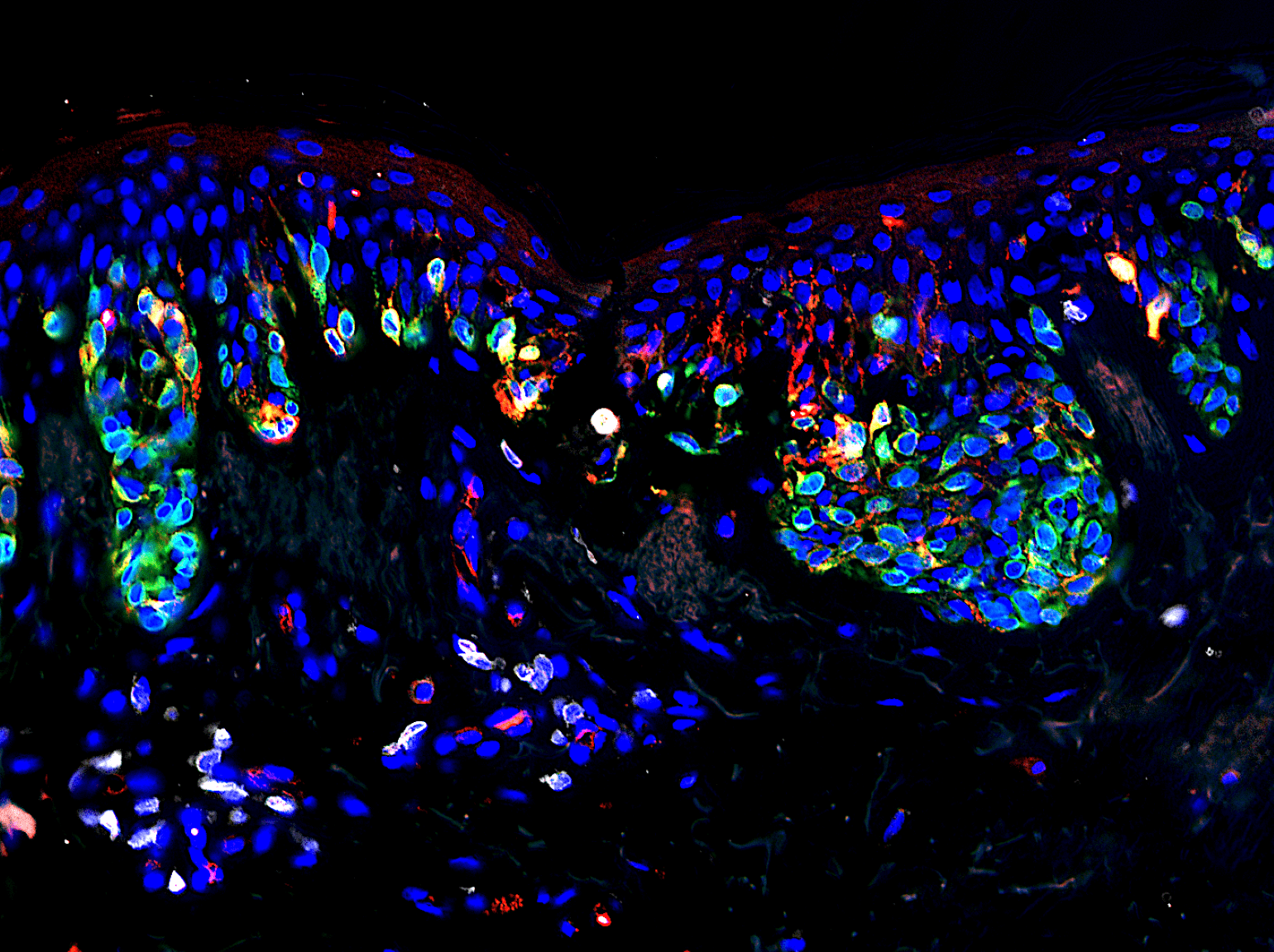
Figure 1
T cells reside in a melanocytic nevus (mole). gp100 (red), MART-1 (green) and CD3+ T cells (white). Image courtesy of Erik Schiferle.
Regulatory T Cells and Nevus Rejection
Ordinarily, CD4+ T helper 1 cells in nevi are held in check by regulatory T cells (Tregs), which prevent rejection of nevi. That action was lost when nevi were transferred into mice.
The researchers confirmed the role of Tregs in their model. Using a low dose of human interleukin-2, they bolstered Treg function in nevi that had been newly transplanted into mice. Five of six xenografts that had IL-2 administered remained stable, then were rejected once IL-2 was stopped.
Similar results were observed after administration of a human leukocyte antigen class II–blocking antibody, which blocks CD4+ T helper 1 cell function.
Therefore, the next important step could be to develop a localized or topical agent for application to nevi that would suppress Tregs.
Mice With Rejected Nevi Are Protected From Melanoma
The researchers injected human melanoma cells into sites on the mice where nevi had been rejected. Melanoma growth and metastasis were significantly blunted at those sites compared with control sites where melanoma cells were implanted into normal human skin.
Therapeutic Implications
Activation of CD4+ T helper 1 cells residing in nevi is a novel strategy to prevent progression to melanoma in high-risk individuals. It may also enhance the effects of immunotherapies for the treatment of melanoma.
view original journal article Subscription may be required
Learn more about the Mass General Cancer Center
Refer a patient to the Mass General Cancer Center