Pharmacological Innovations in Ophthalmology: The Development of Photodynamic Therapy and Anti-Vascular Endothelial Growth Factor
Key Findings
- Mass Eye and Ear celebrated its 200th anniversary in 2024, a year also marking 25 years since the introduction of photodynamic therapy (PDT) and the identification of vascular endothelial growth factor (VEGF) as a therapeutic target for retinal disorders
- In the early 1990’s, the field of retina was largely focused on refining surgical techniques, and treatment options for age-related macular degeneration (AMD) were still extremely limited
- Joan W. Miller, MD, looks back at how a skin cancer treatment inspired her and a team of researchers to develop a more selective and effective treatment for choroidal neovascularization, and how that same team worked together to identify the key role of VEGF in ocular neovascularization, leading to the development of anti-VEGF therapies
This article was co-written by Lauren Granata Carollo, PhD, a science writer at Harvard Ophthalmology/Mass Eye and Ear.
Subscribe to the latest updates from Ophthalmology Advances in Motion
Mass Eye and Ear celebrated its 200th anniversary in 2024, a year also marking 25 years since the introduction of photodynamic therapy (PDT) and the identification of vascular endothelial growth factor (VEGF) as a therapeutic target for retinal disorders.
Where We Started: Advances in Surgery Filled One Gap but Left Another
In the decades since the first vitrectomy was described in 1969, major advances have been made in the treatment of vitreoretinal diseases. At the time Joan Miller, MD, finished her vitreoretinal fellowship in 1991, the field of retina was largely focused on refining surgical techniques. New methods were improving outcomes for patients with retinal diseases such as retinal detachment, diabetic retinopathy, macular hole, and epiretinal membranes, but treatment options for age-related macular degeneration (AMD) were still extremely limited.
The treatment for neovascular AMD (nvAMD) was laser photocoagulation, but outcomes were disappointing for both patient and physician. The laser directed at choroidal neovascularization (CNV) also destroyed the overlying neural retina, and the treated CNV almost always recurred. When CNV was located underneath the fovea, the center of the macula where eyesight is sharpest, treatment led to immediate vision loss and a blind spot. Clearly, there was a need for more selective and effective treatments.
Luckily, a series of developments opened the door for more effective treatments for several ocular pathologies, including neovascular AMD (nvAMD), ushering in an era of pharmacological therapies in retina.
Developing Photodynamic Therapy: From Tumors to CNV
Photodynamic therapy (PDT) had previously been investigated as a treatment for cancers, including intraocular tumors. PDT works by introducing a light-activated photosensitizer to a target tissue and then activating the photosensitizer via a light source at a specific wavelength. PDT of experimental tumors was found to also cause vascular injury, suggesting that it might be effective in treating neovascular tissue. However, previous investigations of PDT in experimental CNV had not demonstrated sufficient selectivity. Ideally, PDT would damage CNV without causing injury to retinal vessels, normal choroidal vessels, or the neural retina.
In 1991, Dr. Rox Anderson at Massachusetts General Hospital was leading the clinical trial for PDT using liposomal benzoporphyrin derivative (BPD), or verteporfin, for metastatic skin cancer. Drs. Evangelos Gragoudas and Tayyaba Hasan, working with fellows Drs. Ursula Schmidt-Erfurth and Wendall Bauman, were also performing studies using BPD to treat experimental choroidal melanoma in rabbits. With a clinical trial already underway using verteporfin PDT for treatment of skin cancers, there was a path forward for PDT of nvAMD if sufficient selectivity could be achieved.
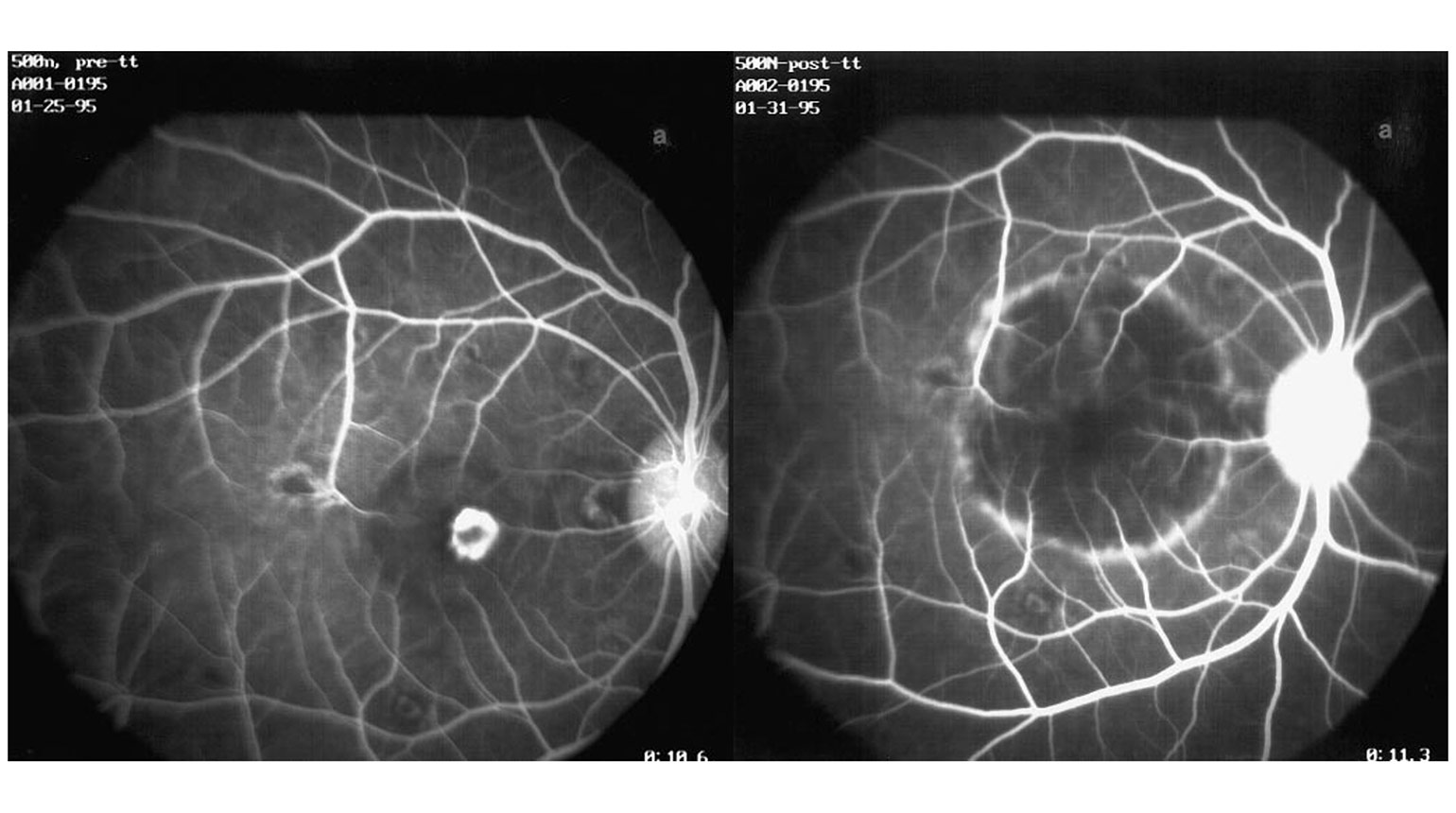
Figure 1
Early-phase fluorescein angiogram of the non-human primate CNV model before (left) and 24 hours after PDT (right), showing an absence of angiographic perfusion and leakage from the CNV after PDT.
(Image adapted from Husain D, Kramer M, Kenny AG, Michaud N, Flotte TJ, Gragoudas ES, Miller JW. Effects of Photodynamic Therapy Using Verteporfin on Experimental Choroidal Neovascularization and Normal Retina and Choroid up to 7 Weeks after Treatment. Invest. Ophthalmol. Vis. Sci. 1999;40(10):2322-2331.)
Gathering Materials for PDT
To explore PDT for nvAMD, Dr. Miller began doing experiments using a nonhuman primate laser injury model to simulate human CNV. To successfully perform these experiments, she would need both BPD and a modified laser. The table top laser at Mass General used to treat experimental ocular tumors provided only limited visualization of ocular structures and did not have the proper range of spot size and power needed to treat CNV. Dr. Miller worked with David Dewey and George Marcelino at Coherent Inc. to build a laser prototype. The 690 nm laser had a slit lamp delivery system that could produce a larger spot size with lower power for PDT. The original BPD photosensitizer, provided by QLT Phototherapeutics, differed from the clinical formulation in that it had to be dissolved in dimethyl sulfoxide; the team would later utilize the liposomal formulation of verteporfin being used in dermatologic clinical trials.
Refining Irradiance Levels
The irradiance level used for skin cancer trials and in experimental choroidal melanoma was low, at only 150 mW/cm2. At this low irradiance, treatment times for CNV were as long as 16 minutes, and it was difficult to focus the laser on a macular lesion for that sustained period of time. Dr. Miller recognized that treatment at the low irradiance would never be practical and proceeded to test higher irradiance levels up to 1800 mW/cm2. Although experts in the field were adamant that higher irradiance would cause pain and other side effects, Dr. Miller was reassured that the heat sink properties of the choroid would protect the retina and choroid.
Dr. Miller and team performed extensive experiments testing photosensitizer dose, light irradiance and fluence, and the timing of laser light application after photosensitizer infusion. These experiments were successful, achieving CNV closure without complications, even at high irradiance levels. When PDT was tested in normal retinas, treatments could be titrated to avoid significant damage to the outer retina. Ultimately, Dr. Miller and her team determined that the experimental parameters of 6 mg/m2 verteporfin dose, irradiance of 600 mW/cm2, and fluence of 50 J/cm2 were ideal for achieving CNV closure in preclinical models while avoiding significant damage to the outer retina.
Based on the successful preclinical studies, Dr. Miller and clinical investigators designed phase 1/2 clinical trials to evaluate the safety and short-term efficacy of verteporfin PDT for subfoveal CNV in patients with AMD and other conditions (including pathologic myopia and angioid streaks). The phase 1/2 trials used an irradiance of 600 mW/cm2, and 2 doses of verteporfin (6 or 12 mg/m2), infusion over 5 or 10 minutes, and varied time of laser light application (10, 15, 20, or 30 minutes after start of infusion) and fluence (50 to 150 J/cm2). Dr. Miller performed the first patient treatment using verteporfin PDT at Mass Eye and Ear in March 1995 and the phase 1/2 trial further refined the treatment parameters.
Following this success, two Phase 3 clinical trials—Treatment of Age-Related Macular Degeneration Using Photodynamic Therapy (TAP)—were carried out in the U.S. and Europe. Over 600 patients with subfoveal CNV secondary to AMD were enrolled across 22 sites with randomization 2:1 of treatment to placebo controls. The primary endpoint, avoiding vision loss of 15 letters or more at one and two years, was met in both trials. Another Phase 3 clinical trial, Verteporfin in Photodynamic Therapy (VIP), found decreased risk of vision loss at one and two years in patients with subfoveal CNV caused by pathologic myopia. Verteporfin PDT (Visudyne®) was first approved for use in Switzerland in December 1999, and additional approvals followed in the United States (April 2000), the European Union (July 2000), and Japan (2003), making it the first pharmacological treatment for nvAMD. At the time, the launch of Visudyne® was the most successful introduction of a drug in ophthalmology.
Subsequent development of anti-VEGF agents, discussed in the following sections, has largely supplanted PDT in treating nvAMD. However, PDT is still useful for some patients with nvAMD, as well as for chronic central serous choroidopahty, and work by Dr. Gregg Kokame and others has demonstrated the effectiveness of PDT for polypoidal choroidal vasculopathy (PCV), a subtype of neovascular AMD, with clinical trials supporting these observations.
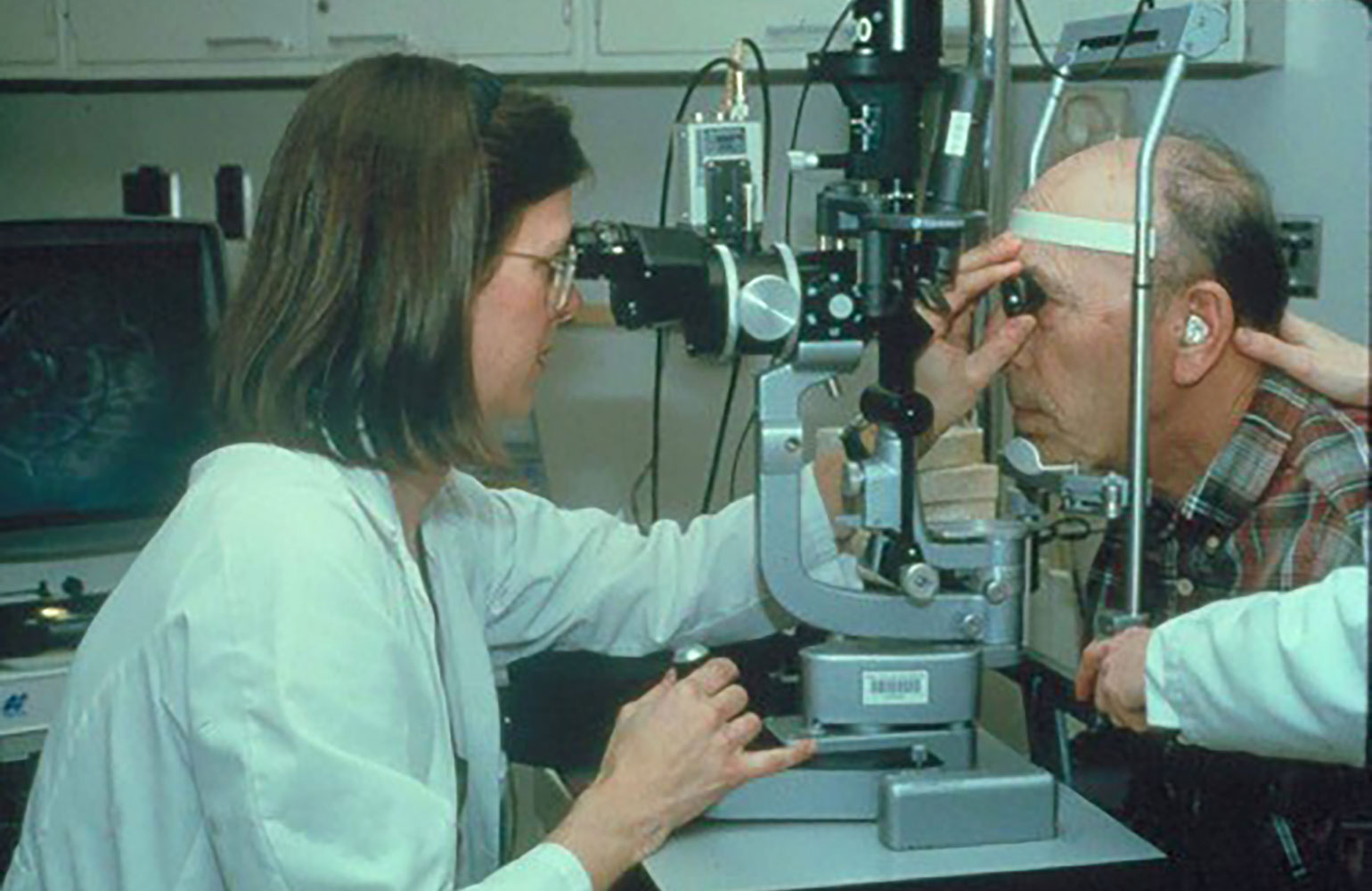
Figure 2
Dr. Miller treating a patient with verteporfin PDT.
(Image adapted from Miller JW, Ophthalmology Retina 2019;3:900-909 and reprinted with permission from the American Academy of Ophthalmology.)
The Road to Discovering the Mechanism of Ocular Neovascularization
At the same time, in the early 1990s, a confluence of researchers, including Dr. Miller, were investigating the drivers of pathologic ocular neovascularization, a major cause of vision loss in AMD and other retinal diseases. The theory that there must be a “factor in the retina capable of affecting the growth of vessels” and “associated with the metabolism of the retinal tissue” was postulated by I.C. Michaelson as early as 1948. This unknown “Factor X” would elude scientists for decades to come.
In 1971, Dr. Judah Folkman, known as the “Father of Angiogenesis,” was the first to propose that angiogenesis was required for tumor growth and metastasis. Based on these observations he pursued drugs that were “anti-angiogenic” as a revolutionary treatment for cancers. These concepts arising out of tumor research were applicable to neovascular ocular disease, and the search for Factor X continued. In the 1980s, Drs. Patricia D’Amore, Bert Glaser, Arnall Patz, and others identified angiogenic fibroblast growth factors 1 and 2 (FGF-1 and FGF-2) in the retina and vitreous as potential candidates for Factor X. While important angiogenic factors, FGFs were ruled out on the basis that they were not secreted.

Figure 3
Dr. Judah Folkman, known as the “Father of Angiogenesis.”
(Photo courtesy of Boston Children’s Hospital Archives, Boston, Massachusetts)
Discovery of VEGF: The Elusive “Factor X”
Harold Dvorak, MD, at Harvard Medical School, identified a secreted permeability factor, vascular permeability factor (VPF) in 1983, observing that it was 50,000 times more potent than histamine. In 1989, Napoleone Ferrara, MD, at Genentech, identified the same secreted factor, recognizing its angiogenic properties and named it vascular endothelial growth factor (VEGF). VEGF had two properties that aligned with what was assumed of the potential Factor X: it was a secreted endothelial cell mitogen, and it was regulated by hypoxia.
In Boston, two groups were investigating VEGF and ocular angiogenesis. Dr. Patricia D’Amore and her graduate student, David Shima, along with Drs. Anthony Adamis, Gragoudas and Miller, led the efforts at Boston Children’s Hospital and Mass Eye and Ear; Drs. Lloyd Paul Aiello, and George King, along with Drs. Eric Pierce and Lois Smith led the efforts at Joslin Diabetes Center and Boston Children’s Hospital.
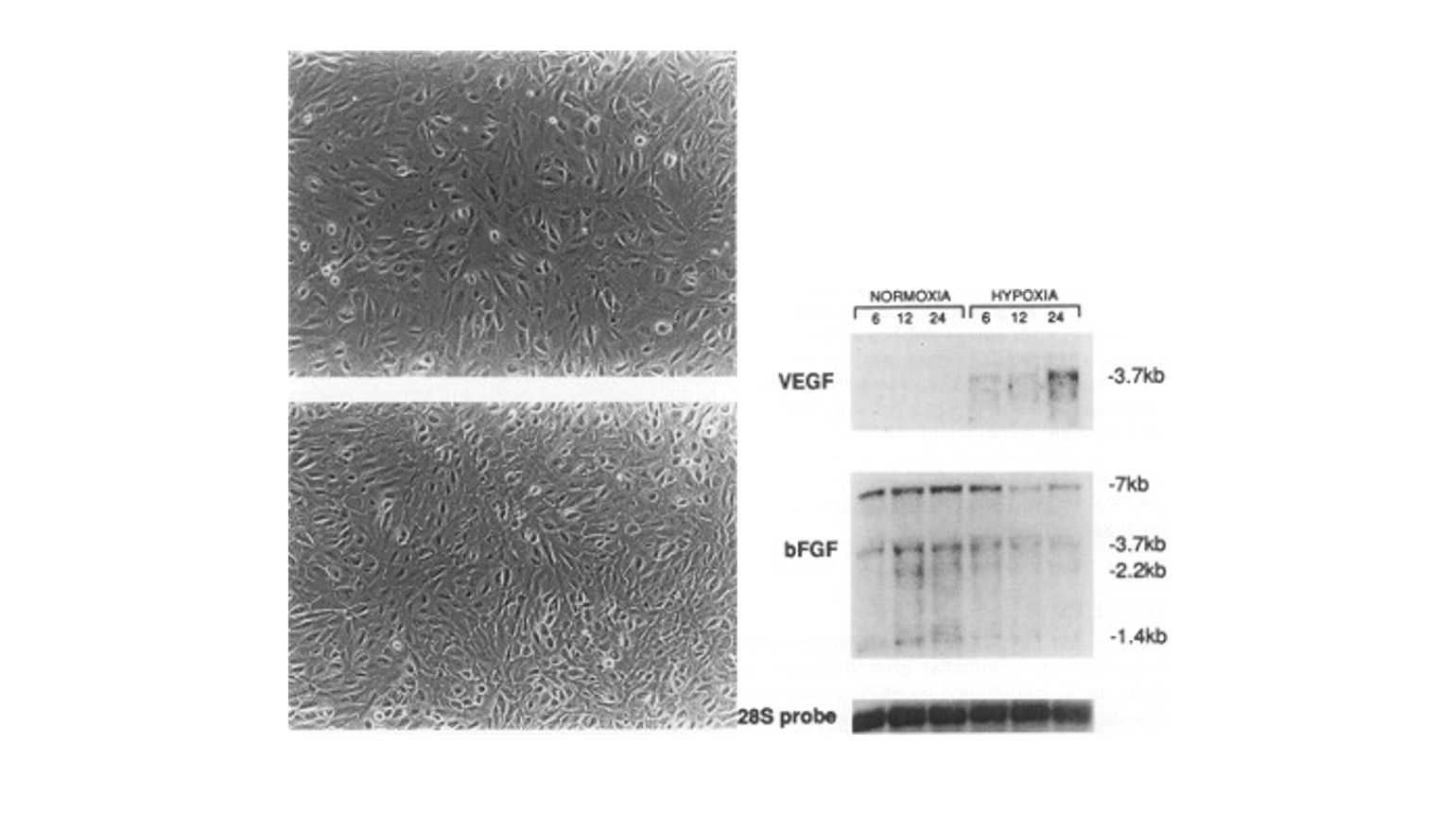
Figure 4
Human retinal pigment epithelium grown under normoxic (top left) and hypoxic (bottom left) conditions, and VEGF mRNA expression after 6, 12, and 24 hours of hypoxia (right).
(Image adapted from Shima DT, Adamis AP, Ferrara N, Yeo KT, Yeo TK, Allende R, Folkman J, D'Amore PA. Hypoxic induction of endothelial cell growth factors in retinal cells: identification and characterization of vascular endothelial growth factor (VEGF) as the mitogen. Mol Med. 1995.)
Preclinical Validation of VEGF as a Target for Therapy
First, Drs. Adamis, D’Amore, and Shima demonstrated that VEGF was secreted by the retinal pigment epithelium (RPE) in culture, and was responsive to hypoxia. At the same time, Drs. Miller and Adamis found that VEGF levels were undetectable in aqueous fluid from normal eyes of nonhuman primates, but rose quickly as retinal ischemia and iris neovascularization developed following laser-induced retinal vein occlusion. This was the first demonstration of a correlation between VEGF protein levels and ocular angiogenesis in vivo. Drs. Miller, Adamis, and D’Amico demonstrated a similar correlation in patients in a small study. This was confirmed by a pivotal study in a large number of patients published by Drs. Aiello and King’s team demonstrating that VEGF protein in aqueous or vitreous was elevated in the setting of retinal ischemia, active proliferative diabetic retinopathy, and iris neovascularization, and decreased following panretinal laser photocoagulation.
Drs. Miller, Adamis and Gragoudas worked with Dr. Napleone Ferrara to investigate the effect of anti-VEGF in the same non-human primate model of retinal ischemia and iris neovascularization using intravitreal injection of an anti-VEGF antibody (the precursor of bevacizumab). The antibody completely prevented neovascularization, proving that VEGF was required for neovascularization. They then demonstrated that injection of VEGF into normal non-human primate eyes led to iris neovascularization and neovascular glaucoma, mirroring pathology observed clinically. An antibody fragment, rhuFab VEGF (the precursor to ranibizumab), also provided by Genentech, was next shown to be effective in the laser-induced CNV model, providing support for clinical investigation in patients with nvAMD. The combined efforts of all these groups demonstrated, in animal models and in patients, that VEGF was required and sufficient for neovascularization; they had found the elusive Factor X.
Anti-VEGF Therapies Enter Clinical Trials for Ophthalmic Applications
Anti-VEGF therapy seemed primed for clinical trials in ophthalmology by the mid-1990’s, but Genentech was more focused on cancer trials. It took a partnership of former Genentech employees, Marty Glick and John McLaughlin, along with Drs. David Guyer, Anthony Adamis and Samir Patel to found Eyetech Pharmaceuticals, licensing anti-VEGF aptamer technology from Gilead Sciences to begin clinical trials in ophthalmology. Pegaptanib, the anti-VEGF aptamer, was successful in phase 2 and later phase 3 clinical trials, slowing and limiting vision loss associated with nvAMD, and received FDA approval in December 2004. This was the first time an antiangiogenic therapy demonstrated efficacy in ophthalmology. While the vision results with pegaptanib treatment were comparable to those of treatment with verteporfin PDT, pegaptanib showed benefit across a wider range of nvAMD subtypes.
Genentech, spurred on by the early results of pegaptanib, developed their own anti-VEGF agents for ophthalmology. The company developed a full-length anti-VEGF antibody, bevacizumab, administered intravenously, which was approved for use by the U.S. FDA in 2004 for metastatic colon cancer. For intravitreal administration in ocular indications, a smaller anti-VEGF antibody fragment, rhuFab (ranibizumab) was developed. In the pivotal phase 3 MARINA and ANCHOR trials, the primary endpoint of preventing moderate vision loss was achieved, but ranibizumab also impressively showed improved visual acuity at one year and two years in patients with nvAMD. Presented for the first time at a meeting in 2005, this was the first demonstration of sustained vision improvement in nvAMD with an anti-VEGF therapy. Based on these studies, ranibizumab was approved for treatment of nvAMD by the FDA in June of 2006.
Comparing AMD Treatment Trials
At the same meeting where the dramatic ranibizumab results were presented, Dr. Philip Rosenfeld presented a single case of a patient with nvAMD treated off label with intravitreal bevacizumab who showed improvement in retinal thickness on optical coherence tomography (OCT). This suggested that, despite previous doubts, the full-length antibody was able to penetrate the retina and demonstrate biological activity. This surprising finding, in addition to the exciting phase 3 results of ranibizumab, generated enthusiasm for full-length anti-VEGF antibodies as well as the antibody fragments for ocular indications.
Although it had not been formally studied or approved for AMD, and Genentech could not legally promote its off-label use, ophthalmologists were eager to try bevacizumab and its off-label use became widespread across the U.S. A National Institutes of Health-funded study was undertaken by Dr. Daniel Martin, the Comparing AMD Treatment Trials (CATT), which demonstrated the safety and efficacy of bevacizumab compared to ranibizumab at one year and two years of follow up.
Today: Options for Anti-VEGF Therapies
Today, many anti-VEGF agents have been approved by the FDA and other agencies internationally for nvAMD and other conditions. By order of approval date, pegaptanib (Macugen®), ranibizumab (Lucentis®), aflibercept (Eylea®), and brolucizumab (Beovu®) are approved for nvAMD. Bevacizumab (Avastin®) is indicated for tumor treatment but used off label for nvAMD. In 2022, faricimab (Vabysmo®) received FDA approval for the treatment of nvAMD and diabetic macular edema (DME). In 2023, aflibercept HD (Eylea HD), a higher, 8mg dose was approved for treatment of nvAMD, DME, and diabetic retinopathy. With the success of these anti-angiogenic agents, many biosimilars have been approved or are in development for the treatment of patients with nvAMD and other eye diseases. Biosimilars for ranibizumab, aflibercept, and bevacizumab are listed in Table 1.
| Ranibizumab | Aflibercept | Bevacizumab |
| Byooviz (Samsung Bioepis, Biogen)* | Yesafili (Biocon Biologics)* | Mvasi (Amgen/Allergan)# |
| Cimerli (Coherus)* | Opuviz (Samsung Bioepis, Biogen)* | Zirabev (Pfizer)# |
| Ximulci (EU; Xbrane) | Pavblu (Amgen)* | Alymsys (Amneal)# |
| CKD-701 (Chong Jun Dang) | Enzeevu (Sandoz)* | Vegzelma (Celltrion)# |
| GNR-067 (Generium Pharmaceutical) | Ahzantive (Formycon AG)* | Avzivi (Bio-Thera Solutions)# |
| OT-701 (Ocumension/Santen) | ALT-L9 (Alteogen) | Oyavas (Clonmel Healthcare) |
| PDP807 (Polus) | MYL1710 (Momenta Pharmaceuticals/Mylan) | Aybintio (Samsung Bioepis) |
| QL 1205 (Qilu Pharmaceutical) | OT-702 (LY09004) (Ocumension) | Onbevzi (Samsung Bioepis) |
| R30 (Shanghai United Cell Biology) | CT-P42 (Celltrion) | Abevmy (Biocon Biologics) |
| SCT510A (Sinocelltech) | AVT-06 (Alvotech) | ONS-510 (Outlook Therapeutics) |
| Razumab (India; Intas Pharmaceuticals) | SCB-420 (Clover Biopharmaceuticals) | |
| Ranizurel (India; Reliance Life Sciences) | GBS-012 (Gene Techno Science) | |
| Ranieyes (India; Lupin) |
Bold type indicates FDA approval
* Indicated for nvAMD
# Indicated for oncology
Although anti-VEGF therapy has revolutionized treatment for nvAMD, new agents can show new risks. For example, after the FDA approval of brolucizumab in 2019, reports followed of retinal vasculitis with significant and permanent vision loss, limiting adoption of the drug. Additionally, the high treatment burden associated with frequent injections spurred investigations of alternative therapies. These include sustained release delivery systems, longer-acting agents, combination therapy, and gene therapy.
Polymer-based sustained-release systems must maintain the fragile protein structure of anti-VEGF agents, but this proved difficult to achieve. Genentech, a member of the Roche group, turned to the ranibizumab port delivery system, an implantable and refillable device, which can deliver active agent over the course of 6 months. The port delivery system aims to maintain the anti-VEGF treatment benefits while limiting the burden of repeated injections. The first-of-its-kind system, Susvimo (ranibizumab injection), obtained FDA approval in 2021 for treatment of nvAMD after showing vision gains were equivalent to monthly ranibizumab injections. However, technical difficulties after implatationrequired the temporary suspension of new implantations, and clinical trials were paused. After changes to the implant and additional testing, Susvimo returned to the market in 2024.
Longer-acting anti-VEGF agents, such as brolucizumab (Beovu), a humanized monoclonal single-chain variable fragment, aim to reduce the number of injections required to sustain vision improvements. Brolucizumab binds and inhibits VEGF with high affinity and can be effective with 8- or 12-week dosing intervals, but carries an increased risk of inflammation, which remains a consistent safety concern. Other treatments promoted as longer-acting include high-dose aflibercept (Eyelea HD) and faricimab (Vabysmo®), a combination anti-VEGF/anti-angiopoietin-2 treatment. While clinical trials show vision improvements are possible with fewer injections, in clinical practice, injection frequency is typically guided by individual patient responses, and can still require monthly dosing. Without further comparison trials, the question remains whether these agents are truly longer-acting.
Gene therapies hope to provide a one-time treatment to promote expression of anti-VEGF proteins over the course of a lifetime. Early trials of gene therapies, such as Adverum Biotechnologies’ ADVM-022, were problematic because of adverse events, especially inflammation common with intravitreal adenoviral (AV) and adeno-associated viral (AAV) injections. Regenxbio is developing a promising AAV candidate, RGX-314, delivered subretinally or suprachoroidally, which limits the risk of inflammation.
The continued development of treatments that reduce the financial, physical, and emotional burden on patients and families remains an unmet need. Treatments that require fewer clinical visits and fewer injections, and limit invasive procedures would be tremendously valuable.
Before these novel pharmacological advancements for the treatment of ocular diseases were brought on the scene, patients with nvAMD and other retinal conditions had very few options, and outcomes were not encouraging. The development of PDT, and then the discovery of VEGF as “Factor X” leading to therapeutics targeting VEGF, revolutionized the field of ophthalmology and provided a strong foundation for future treatments.
Drs. Joan W. Miller, Evangelos S. Gragoudas, Patricia A. D’Amore, Lloyd Paul Aiello, George L. King, Anthony P. Adamis, and Napoleone Ferrara were awarded the 2014 Antonio Champalimaud Vision Award for the development of anti-angiogenic therapy for retinal disease. As Mass Eye and Ear celebrates 200 years, the revolutionary discoveries that have transformed the possibilities in eye care are continuing to be recognized as truly remarkable achievements.

Figure 5
Winners of the 2014 Antonio Champalimaud Award Vision Award. Drs. Napoleone Ferrara, Evangelos Gragoudas, Joan Miller, Anthony Adamis, George King, Patricia D’Amore, and Lloyd Paul Aiello. Courtesy MEEI.